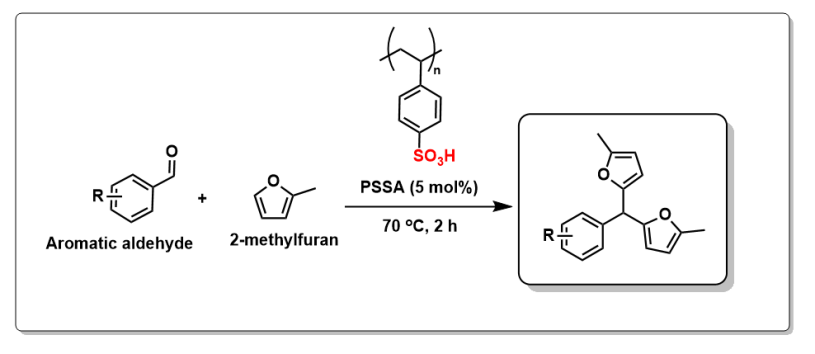
Following the application of polystyrene sulfonic acid (PSSA) as a catalyst in the synthesis of a biofuel precursor from furfural and 2‑methylfuran, we explored the scope of Friedel–Crafts reactions between aromatic aldehydes and 2‑methylfuran catalysed by PSSA.
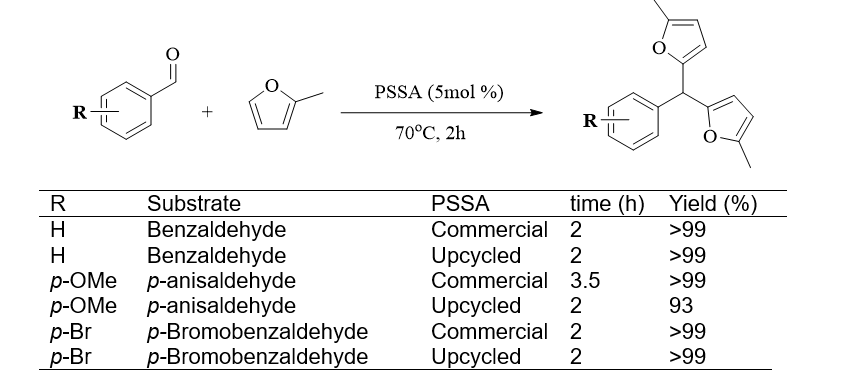
Our results have been published in RSC Sustainability. The study began with the optimisation of the Friedel–Crafts reaction using aromatic aldehydes bearing different functional groups and substitution patterns, employing a commercially available sample of PSSA as the catalyst. Under the optimised conditions, the reaction proved highly efficient, affording the corresponding products in nearly quantitative yields for most substrates investigated. We then compared Friedel–Crafts reactions involving the α‑aryl aldehydes, benzaldehyde, p-anisaldehyde, and p-bromobenzaldehyde using upcycled PSSA and commercial PSSA. Overall, the upcycled PSSA demonstrated comparable catalytic efficiency to the commercial PSSA under the tested conditions. All products were obtained using upcycled PSSA with similar reaction times and yields. However, twice the amount of upcycled PSSA was required, reflecting its lower sulfonation degree (44%) compared with commercial PSSA (90%). Interestingly, in the case of p-anisaldehyde, the upcycled PSSA led to a faster reaction, although with a slightly reduced yield.
All experimental protocols and characterisation data are freely available in our library of synthesised compounds using upcycled PSSA. In addition, a step‑by‑step video tutorial demonstrating how to set up Friedel–Crafts reactions with 2‑methylfuran (using furfural as a model aldehyde) is available on our RECOMPENSE YouTube channel.